Serving you since 1994
Serving you since 1994

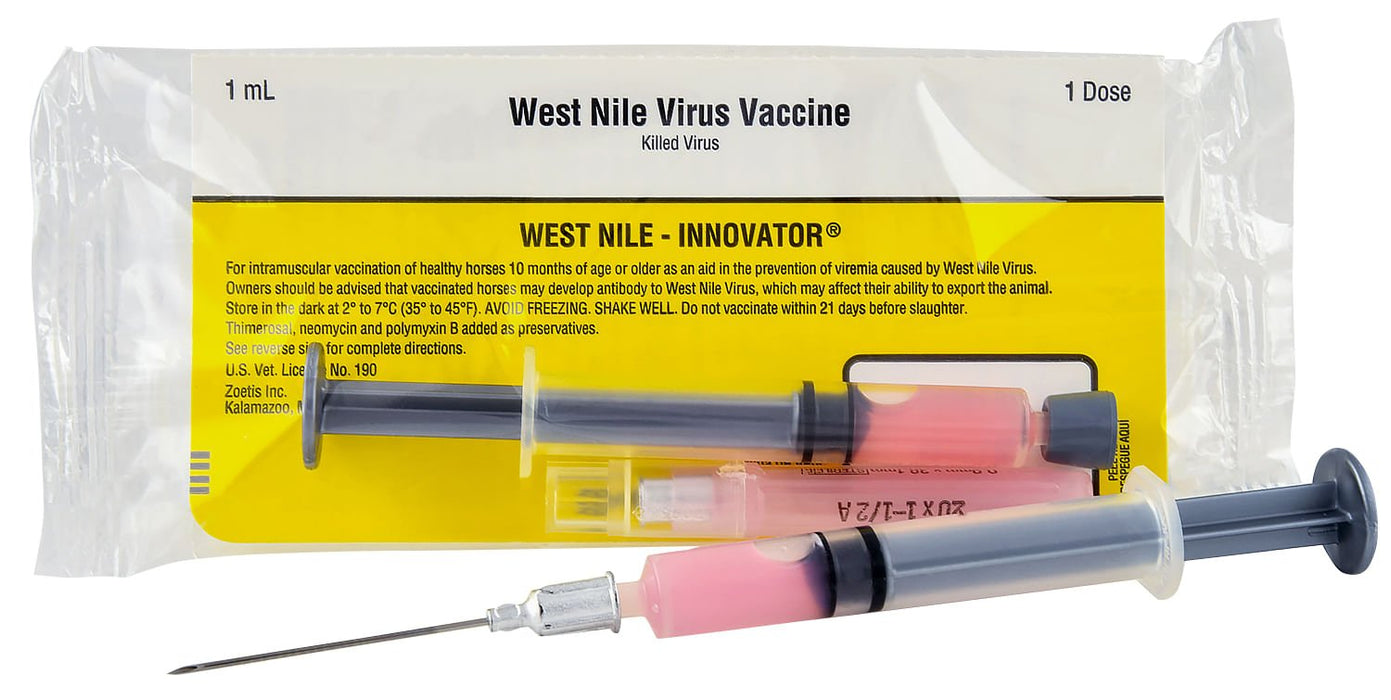
For the vaccination of healthy horses as an aid in the prevention of viremia caused by West Nile Virus.
Single dose or 10 dose vial.
Single dose includes needle and pre-loaded syringe.
A Valid veterinarian prescription is required to purchase this item in the state of Minnesota. Please contact your veterinarian for a prescription.
All vaccines are NON-RETURNABLE and NON-REFUNDABLE.
{"one"=>"Select 2 or 3 items to compare", "other"=>"{{ count }} of 3 items selected"}