Serving you since 1994
Serving you since 1994

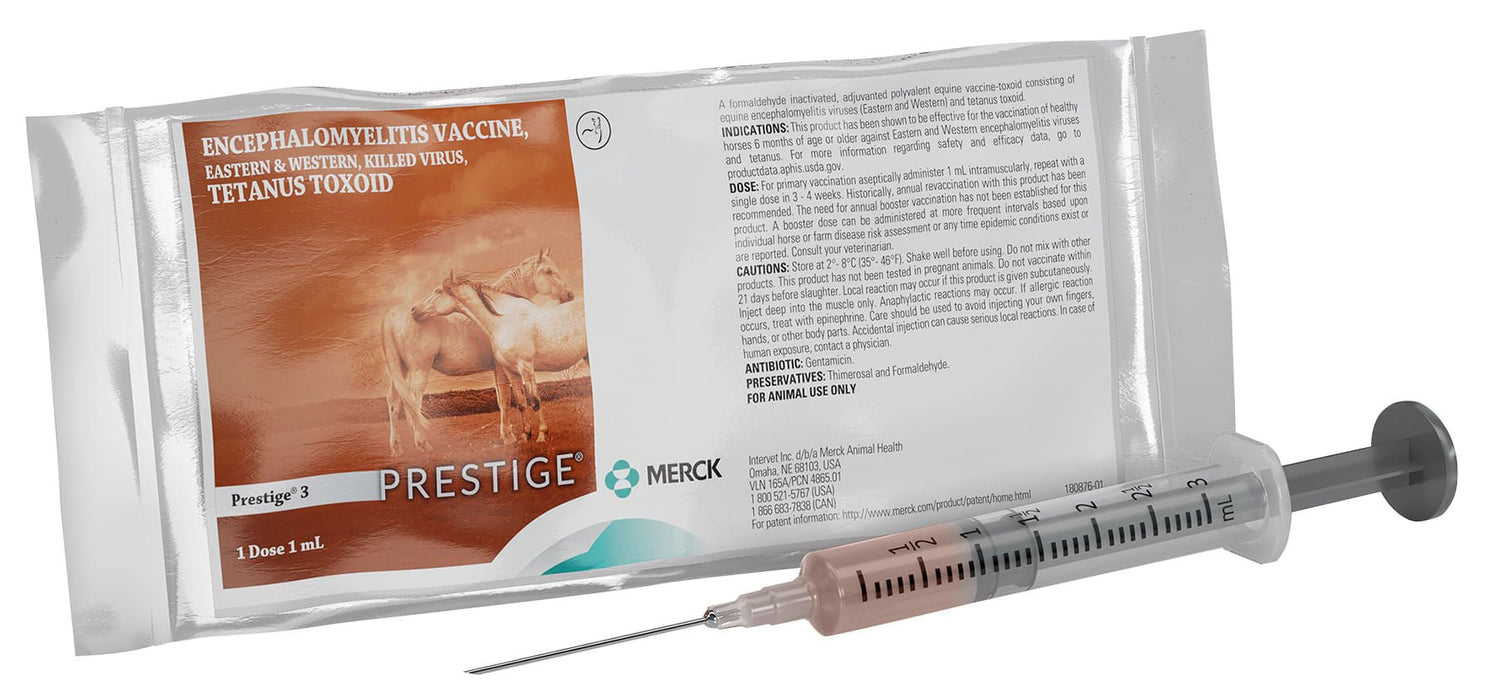
Prestige 3 is a horse vaccination that has been shown to be effective in healthy horses 6 months of age or older. Prestige 3 fights against encephalomyelitis viruses and tetanus.
A Valid veterinarian prescription is required to purchase this item in the state of Minnesota. Please contact your veterinarian for a prescription.
All vaccines are NON-RETURNABLE and NON-REFUNDABLE.
{"one"=>"Select 2 or 3 items to compare", "other"=>"{{ count }} of 3 items selected"}